Physics ss1 term 3
Physics ss1 term 3
Physics ss1 term 3
FIELD FORCE
CONTENT
- The Concept of Fields
- Types of Fields
- Properties of a Force Field
- Acceleration Due to Gravity
- Determination of Acceleration Due to Gravity
- The Shape and Dimension of the Earth
The Concept of Fields
A field is a region under the influence of some physical agencies such as gravitation, magnetism and electricity.
Types of Fields
There are two types of field:
(i) Vector field
(ii) Scalar field.
Vector Fields
A vector field is that field which is usually represented by lines of force; while a scalar field is that field that is not represented by lines of force.
Examples of vector fields include gravitational field, magnetic field and electric field.
Examples of scalar fields include regions with distribution of temperature, density, etc.
(i) Gravitational Field
Gravitational field is a region of space or a force field surrounding a body that has the property of mass. In this region, any object that has mass will experience a force of attraction, called gravitational force.
Gravitational force is responsible for the fact that any object thrown up must definitely fall back. This force of gravity pulls every object towards the centre of the earth. That is to say, gravitational force causes a body which is not in contact with the earth to fall to the ground. This therefore means that the earth exerts an attractive force on every object either on it or near it.
Similarly, two objects of different masses exert equal and opposite forces of attraction on each other.
The radial field near a planet (e.g, earth) is shown below:
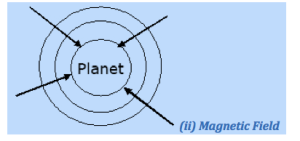
Magnetic field is a region around a magnet where it exerts force on other magnets. It is also a region where magnetic force is felt.
The patterns of the magnetic lines of force are shown below:
Magnetic Field Patterns
- Field of a bar magnet:
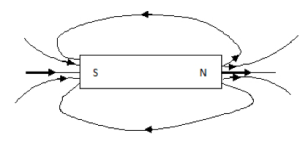
2. Attraction between unlike poles: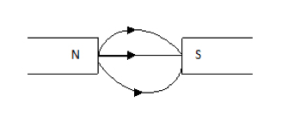
3. Repulsion between like poles:
(iii) Electric Field
An electric field is a region around an electric charge where it exerts force on other charges. It is a field where an electric influence is felt.
The patterns of the electric lines of force are shown below:
- Isolated positive and negative charge field lines:
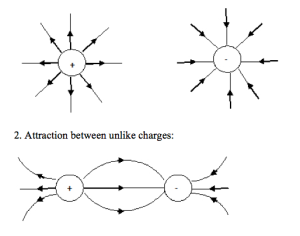
EVALUATION
- What is a field?
- State the two types of field.
- List the examples of vector field.
- What is neutral point?
Properties of a Force Field
(i) Properties of Gravitational Field
(a) The lines of force are directed towards the centre of the planet; hence, it is a radial field.
(b) The gravitational force field (field strength) ‘g’ at a point is the force per unit mass placed at that point. i.e, g=Fm in N/kg but the S.I unit is m/s2
(c) Any force acting on a body falling towards the centre of the earth is given by F = mg
(d) Gravitational field is a vector quantity.
(ii) Properties of Magnetic Field
(a) Direction: When a magnet is freely suspended, it comes to rest in the South-North direction of the earth.
(b) Attraction: A magnet has the ability to attract magnetic materials e.g, steel, iron, etc.
(c) Force: A magnet exerts force on other magnets in such a manner that like poles repel and unlike poles attract.
(d) The inseparable nature of poles on the magnetic dipoles: If a magnet is broken into small pieces, however small it may be, it will still have a North and South Poles. The smallest bit of a magnet is a dipole.
(e) Magnetic lines of force originate from the North pole and terminate at the South pole.
(iii) Properties of Electric Field
(a) Electric lines of force originate from a positive charge and terminate in a negative charge.
(b) Electric lines of force never cross each other.
(c) They repel each other side ways.
(c) They are in a state of tension which tends to shorten them.
(d) The electric field at a point is defined as the force per unit charge placed at that point. i.e, ε=Fq measured in Newton per Coulomb N/C
EVALUATION
- State two properties each of the three vector fields discussed.
- What is the direction of the magnetic lines of force?
- What is the unit of electric field strength?
GENERAL EVALUATION
- Discuss the properties of the magnetic flux.
- Define the electric field strength.
- Itemise the three vector fields.
- Why is electric When a field is represented by lines of force, it is then calledlines of force a vector quantity?
Acceleration Due to Gravity
When an object is dropped from the top of a hill or even a tree, the body moves and increases in velocity until it touches the ground with a velocity of finite value. Such movement is influenced by the earth’s gravitational field. The increase in velocity is therefore due to acceleration due to gravity which is usually represented by ‘g’. The motion of such body under gravity is always described as motion under free fall.
However, when two bodies of different masses are released from a height above the ground level, they do hit the ground at the same time. This is because acceleration due to gravity at a location is the same for all bodies irrespective of their masses and thus reach the ground at the same time.
This constant acceleration is called acceleration due to gravity and has a value of or .
When a body is released from a height so that it falls towards the centre of the earth, ‘g’ is positive; but when a body is thrown upward, it goes against ‘g’ thereby decreasing in velocity until it momentarily comes to rest at the maximum height. For upward movement, ‘g’ is negative.
The equations connecting acceleration due to gravity, ‘g’ are as follows:
For downward movement, v2=u2+2gs and s=ut+12gt2
For upward movement, v2=u2–2gs and s=ut−12gt2
When a body is released from rest at a certain height so that it falls towards the centre of the earth,
For upward movement, s=ut+12gt2
Since, u = 0, 2s = gt2
∴ t2=2sg
Hence, t=2sg−−√
This equation shows that the time to reach the ground does not depend on the mass of the object.
Determination of Acceleration Due to Gravity
The value of ‘g’ could be determined using:
- Formula method: A body is released from a height ‘s’ and the time t is taken; then use s=12gt2 to get the value of ‘g’.
- Simple Pendulum Experiment method: The value of ‘g’ could also be determined using this experiment.
The period T for the oscillation is given by: T=2πlg−−√
By linearizing this formular, we have T2=4π2(lg)
When T2 is plotted against l, the equation is T2=(4π2g)l
- Hence, the slope for such graph is 4π2g
When l is plotted against T2, the equation is l=(g4π2)T2
And the slope for such graph is g4π2
In any case, from the slope, you get the value of ‘g’.
(NB: Educator should carry out the two experiments with the students.)
EVALUATION
- What is the value of acceleration due to gravity?
- What is the mathematical relationship between the period of oscillation T and the length of the string used l in a simple pendulum experiment?
The Shape and Dimension of the Earth
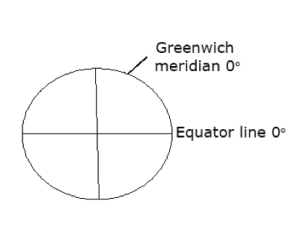
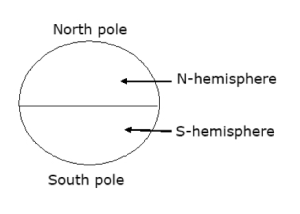
The earth is one of the nine planets in the solar system. It is spherical is shape. It is also divided into two hemispheres – the Northern and Southern hemispheres. There are two major types of lines that run through the earth. They include:
- The latitude lines and
- The longitude lines
The latitude lines are imaginary lines running from the east to the west, north or south of the equator. This means that they increase towards the North or South. Examples are:
- Tropic of cancer
- Tropic of capricon
- Artic circle
- Antartic circle.
- The equator line on zero degree.
The longitude lines are imaginary lines running from the North pole to the South pole, east or west of the Greenwich meridian. They increase towards the east or west. E.g, the Greenwich meridian on zero degree running through Ghana and London.
However, the earth has a radius of approximately 6400km.
EVALUATION
- State two differences between latitude lines and longitude lines.
- Mention two examples of the lines of latitude.
- What is the approximate radius of the earth?
THE PARTICLE NATURE OF MATTER
CONTENT
- The Definition of Matter
- Structure of Matter
- Evidence of the Particle Nature of Matter
- Experimental Evidence of the Particle Nature of Matter
- Explanation of Brownian Motion
- Simple Atomic Structure
- The Nature and Size of Molecules
The Definition of Matter
Matter is anything that has weight and occupies space. Every object or substance is made up of matter. Many of the properties and behavior of substances can best be explained by assuming that all substances are composed of small particles called molecules. The assumption that matter is made up of tiny particles (molecules) which are in constant motion is known as the “molecular theory of matter.”
Structure of Matter
Evidence of the Particle Nature of Matter
(i) Many substances in solid form can easily be crushed to powder form e.g. piece of chalk, lump of clay, charcoal and piece of stone.
(ii) A dry stick or dry wood is easily broken into smaller bits
(iii) Solubility – if you drop a cube of sugar into a cup of water and turn the water, the sugar “disappears”. That is, it dissolves in water.
(iv) If you scrape the surface of a piece of chalk, you will see thousands of very tiny particles flake off and float through the air
(v) If a beam of light (e.g. sunlight) is entering a dusty room through a window, you will observe a chaotic motion of the dust particles in the air.
Experimental Evidence of the Particle Nature of Matter
Experimental evidence of the atomic or molecular nature of matter is the Brownian motion named after the botanist, Robert Brown, who discovered the phenomenon in 1827. While he was observing tiny pollen grains suspended in water under a microscope, Brown noticed that the tiny pollen grains moved about in zigzag paths even though the water appeared to be perfectly still. The here and there by the molecules of water pollen grains were being knocked about which were vigorously moving about.
Brownian motion can be demonstrated by the smoke-cell experiment. The apparatus is shown in the figure below.
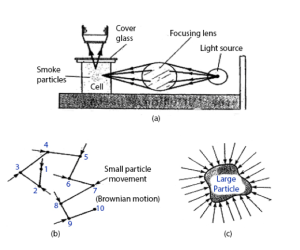
Molecular motion in gas: Brownian (random) motion
Collect some smoke from a smoldering piece of cloth or wood by means of a syringe and introduce it into the cell. Replace the cover quickly and adjust the focus of the microscope until the fine particles come into view clearly. You will observe the smoke particles as black dots which move about irregularly like a (drunkard) drunken man staggering about. The particles dart from one place to another very suddenly, some going out of focus, others coming into focus, but always in motion.
Explanation of Brownian Motion
The irregular movement of the smoke particles is due to the motion of the invisible air molecules which bombard each particle from all sides. The particle is very small and the number of molecules of air hitting one side is not balanced by the number of molecules hitting the opposite side at the same instant. Therefore, the particle moves in the direction of the resultant force and when it moves to another place, the same thing happens. Why can’t the motion of the particles be due to convection? In that case the particles would move upwards continually and not zigzag from side to side.
Brownian motion in liquids can be demonstrated similarly as follows:
Place on a clean microscope slide a few drops of diluted aquadag (finegraphite particles suspended in water) or photopake (a similar suspension used for blacking negatives) and cover the liquid with a cover-slip. Project an image of the slide on a screen using a micro projector so that the particles can be seen. The graphite particles are then seen to be moving about in an irregular manner, thus showing Brownian motion in a liquid. In this case, the irregular motion of the graphite particles is due to their bombardment by the surrounding water molecules which are constantly moving about in different directions. Brownian motion is important for two reasons.
(i) It provides evidence for the existence of the tiny particles of matter called molecules
(ii) It gives evidence that molecules are in a constant state of random motion.
Physics ss1 term 3
Simple Atomic Structure
Definition of an Atom
An atom is the smallest indivisible particle of an element which can take part in a chemical change.

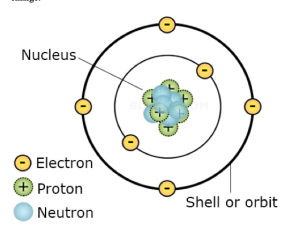
The figure shows the simple structure of an atom. It consists of two parts: The nucleus and the electrons. The nucleus is the heavy portion of the atom and is made up of two types of particles called protons and neutrons. The protons carry a positive charge while neutrons carry no charge. The electrons carry a negative charge and circle in orbits around the heavy nucleus. The numbers of orbits depend on the substance, for example hydrogen has only one orbit while oxygen has two. An electron is very light (about 1 /1840 of the mass of the proton). The negative charge of an electron is equal to the positive charge of a proton and the number of electrons in an atom is equal to the number of protons. The atom is therefore electrically neutral.
The below table summarizes the properties of the elementary particles in the atom of an element.
| S/N | Elementary Particles | Charge (Coulomb) | Mass (kg) | Location in the atom | |
| 1 | Proton | Positively charged (+1.6×10−19) |
(1.67×10−27) | Nucleus | |
| 2 | Neutron | Neutral | (1.67×10−27) | Nucleus | |
| 3 | Electron | Negatively charged (−1.6×10−19) |
(9.1×10−31) | Outermost shell or orbit |
|
| Particle | Charge | Mass (kg) | Relative Mass (amu) | |
| Proton | +1 | (1.6727×10−27) | 1.007316 | |
| Neutron | 0 | (1.6750×10−27) | 1.008701 | |
| Electron | -1 | (9.110×10−31) | 0.000549 (11836) | |
The important points to keep in mind are as follows:
(i) Protons and neutrons have almost the same mass, while the electron is approximately 2000 times lighter.
(ii) Protons and electrons carry charges of equal magnitude, but opposite charge. Neutrons carry no charge (they are neutral).
EVALUATION
- What are the elementary particles contained in the nucleus of an atom?
- Write down their masses and charges.
- Which particle is located in the outermost shell?
Physics ss1 term 3
The Nature and Size of Molecules
All matter is made up of tiny particles called molecules. These molecules themselves are made up of tinier particles called atoms. Both molecules and atom are too tiny to be seen with the naked eyes.
One mole of every substance is believed to contain about 6.02 × 1023 molecules. One molecule of a substance could be found from the combination of two or more elements of that substance.
Definition of Molecule
Hence, we define a molecule as “the smallest unit of matter that is capable of independent existence”. This means that a molecule of a substance could exist alone.
Definition of Atom
An atom is defined as the smallest unit of matter that can take part in a chemical reaction and is not capable of independent existence.
Definition of Element
An element is any substance in which everything could be built up. It is a substance which consists of only one kind of matter and cannot be broken down into anything simpler by any chemical means.
Physics ss1 term 3
EVALUATION
- (a) Define the following terms: (i) Molecule (ii) An atom (iii) An element.
- Which of the above is capable of dependent existence?
- Which of them is capable of independent existence?
GENERAL EVALUATION
- Differentiate an atom from a molecule.
- Highlight the states of matter.
- Enumerate the factors capable of influencing diffusion rate.
- What were the evidences to prove the particulate nature of matter?
THE PARTICLE NATURE OF MATTER
CONTENT
- Molecules
- Definition of Molecules
- Structure, Nature and Size of Molecules
- Some Ideas about Molecular Size
- Estimating the Size of a Molecule
- States of Matter
- The Kinetic Molecular Theory of Matter
- Fundamental Assumptions of the Kinetic Molecular Theory
- Basic Assumptions of the Kinetic Theory of Gases
- Characteristics of the Three States of Matter
- Crystalline and Amorphous Substances
- Crystals
- Non-Crystalline and Amorphous Solids
- Differences between Amorphous and Crystalline Substances
Physics ss1 term 3
Molecules
Definition of Molecules
A molecule is the smallest particle of a substance which can have a separate existence and still retain the properties of that substance.
Structure, Nature and Size of Molecules
(i) Most substances cannot exist by themselves as individual atoms, rather they combine their atoms with themselves or with other atoms to form molecules. Thus a molecule may be made up of similar atoms of the same element or different atoms of two or more elements. For example a molecule of hydrogen is made up of two atoms of hydrogen but a molecule of water consists of two atoms of hydrogen and one atom of oxygen.
(ii) The molecules of any pure substance are identical they have the same structure, the same mass and the same mechanical properties.
(iii) Molecules are formed by atoms combining in simple proportions
(iv) The simplest model of a molecule is that of a rigid sphere (like a small billiard ball) which can move and collide with other molecules or with a wall and exert attractive or repulsive forces on neighboring molecules. The molecular forces decrease as the distance separating the molecules increase.
(v) Molecules are in constant motion. The motion is random (haphazard or zigzag) in liquids and gases but oscillatory or vibrational in solids.
(vi) The size of a molecule is extremely small – of the order of 10-9– 10-10 m. as a result of the small size, molecules cannot be see with the naked eyes or even with the aid of a microscope: Again because of the small size, one gram of an element contains several millions of molecules. For example one gram of hydrogen contains about 1023 molecules.
Some Ideas about Molecular Size
It is difficult to imagine how tiny the diameter of a molecule actually is. Even the numerical value estimated to be of the order of 10-10 m (10-9 cm) is also not easily and accurately conjectured. The following may enable you to have a better idea of the size of a molecule:
(i) If a fine hair is magnified until its thickness is that of a wide street, a molecule in the hair would then look like a speck of dust in the street
(ii) The tip of a pin contains millions of molecules.
(iii) Two grains of hydrogen contain 6 x 1023 molecules.
If the whole population of the world were to count such a huge number of molecules individually at the rate of five molecules per second, it would take 100 years to count all of them.
Physics ss1 term 3
Estimating the Size of a Molecule
You can estimate the size or diameter of a molecule by performing the oil film experiment. The principle of the oil film experiment was discovered by Lord Raleigh in 1890. It was known that certain oils when dropped on the surface of water, would spread to form a circular film with the molecules standing up – right. Lord Raleigh argued that if a drop of oil is placed on top of a water surface, the oil will spread out on top of the water surface until the thickness of the oil film is one molecule thick. He, therefore, used this reasoning to obtain the first estimate of the diameter of a molecule. You can repeat Lord Raleigh’s experiment as follows:
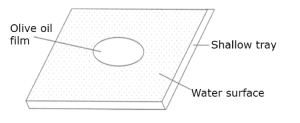
Physics ss1 term 3
(i) Fill a shallow tray with water and allow it to stand until the water is at rest.
(ii) Sprinkle some lycophodium powder lightly on the surface of the water
(iii) Using a graduated pipette with a fine bore, take up a small volume of olive oil and note the reading on the pipette scale
(iv) Drop a very small quantity of the oil on the water surface. Note again the reading on the pipette and obtain the volume of the oil dropped by subtracting the second from the first pipette reading.
(v) Allow the oil to spread, pushing the lycopodium powder outwards and forming a clear thin circular film of oil on thewater surface.
(vi) Measure the diameter of the oil film to the nearest centimeter using a half millimeter scale. Calculate the thickness of the oil film as follows:
Let diameter of oil film = dcm
Let volume of oil drop = Vcm3
Area of oil film = pd 2 = 2 cm2
.’. Thickness of oil film =volumeareaV2=pd2=4Vcm
If you have performed the experiment accurately you should get a value of about 2 × 10-7cm as the thickness of the oil film. Hence the size of an oil molecule is taken as about 2 × 10-7cm
Physics ss1 term 3
States of Matter
Matter exists in three main states, namely solid, liquid and gas.
Solids have fixed shape and volume. They cannot be poured.
Liquids have fixed or constant volumes but they assume the shape of the container. They can be poured.
Gases have no fixed shape, but always occupy the shape of the container. They can be poured.
The Kinetic Molecular Theory of Matter
The theory states that matter is made up of tiny particles called molecules which are in constant motion.
Fundamental Assumptions of the Kinetic Molecular Theory
(i) Matter exists either in solid, liquid or gaseous state.
(ii) All substances consist of molecules, the smallest particle which can exist independently.
(iii) In solids the molecules vibrate about a mean or fixed position. The forces between the molecules are strong and may be attractive or repulsive. All true solids have a crystalline structure in which the atoms are arranged in regular patterns called lattices.
(iv) In liquids the molecules move freely in all directions. In addition to vibrational energy, they have translational energy. The Kinetic energy of the liquid molecules is greater than in solids.
(v) In gases the molecules are in constant motion and are further apart than in solids and liquids. They move at high speeds and have translational, vibrational and in addition rotational energy if the molecules are made of two or more atoms. The attractive or cohesive force is negligible, so, gases are perfectly free to expend and completely fill the vessels containing them. Gas molecules have the greatest Kinetic energy. Because the intermolecular forces are small, the motion of molecules in the gaseous state is linear until collision takes place either with other molecules or with the walls of the container.
Physics ss1 term 3
Basic Assumptions of the Kinetic Theory of Gases
The Kinetic theory of matter has been more completely developed for gases than for solids and liquids. This is because the problems involved are much simpler in the case of gases. The simplest substance to which the theory has been applied is the ideal gas. The fundamental assumptions of the theory are as follows:
(i) Gases consist of many very small particles called molecules, which are like perfectly elastic spheres and are usually in constant random motion.
(ii) Molecules exert no forces on one another except when they collide. Therefore, between collisions with other molecules or with the walls of the container, they move in straight lines.
(iii) Collisions of molecules with one another or with the walls of the container are perfectly elastic. This means that the total Kinetic energy of two molecules before collision is the same as that after collision, and that when a molecule collides with the wall its Kinetic energy is unchanged.
(iv) The duration of a collision is negligible compared with the time between collisions
(v) Molecules are separated by distances which are very large compared with the size of the molecules (or the volume of the molecules is negligible when compared with the volume of the container); they are, however, distributed uniformly throughout the container.
(vi) Any finite volume of the gas contains a very large number of molecules. This assumption is supported by experimental evidence because under standard temperature and pressure (s.t.p), there are about 3 x 1019 molecules per cm3 of any gas.
Characteristics of the Three States of Matter
| S/N | Solids | Liquids | Gases | |
| 1. | Have definite shape | Have no definite shape. They take the shape of their container. |
Have no definite shape | |
| 2. | Have fixed size and volume | Have fixed size and volume | Have no fixed size and volume but spread easily and occupy the volume of their container |
|
| 3. | They don’t move easily | They can move easily | They move faster than liquids |
|
| 4. | The molecules are closely packed and held together by strong intermolecular forces |
Intermolecular distances are greater than that of solids but intermolecular forces are weaker than that of solids |
Intermolecular distances are the farthest and intermolecular forces are weak and negligible |
|
| 5. | They do not mix with other solids |
They may mix or not mix with other liquids |
Mix easily with other gases |
|
| 6. | They are compressible | They are incompressible | They are compressible | |
Crystalline and Amorphous Substances
Solids are usually classified into two groups:
(i) Crystals or crystalline solids;
(ii) Noncrystal or non-crystalline solids;
The difference between crystals and non-crystals is the arrangement of atoms or molecules in the solid.
Physics ss1 term 3
Crystals
Definition of Crystals
A crystal is a piece of solid matter in which the atoms, molecules or ions are arranged in a highly regular repeating pattern called lattice.
Crystal Lattice
The particles in a crystal are arranged in regular 3-dimensional framework or pattern called crystal lattice which repeats over and over again in all directions. The high degree of regularity and order in the arrangement of the molecules is the principal feature distinguishing solids from liquids. Particles in a liquid are jumbled and highly disorganized as they move about. They are even more disorganized in a gas. Examples of common crystals are: sodium chloride, zinc sulphide, chromium, iron and platinum salts.
Physics ss1 term 3
Structure of Simple Crystals
A simple crystal is made up of a huge number of simple basic units or building blocks called unit cells. If you stack these units up and, down, side by side and in all directions, you can build the whole lattice. Unit cells are of three types, giving rise to 3 types of lattice and hence 3 types of
